 ZNG Medical
ZNG Medical
 ZNG Medical
ZNG Medical
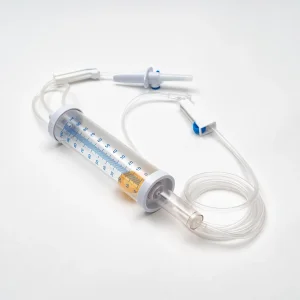 Medical Equipment Infusion Set Burette Infusion Set with Titration Infusion Set
Medical Equipment Infusion Set Burette Infusion Set with Titration Infusion Set
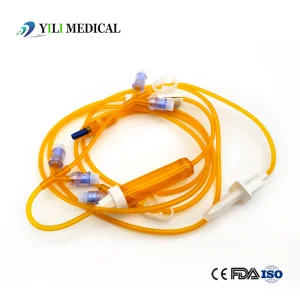 Factory Wholesale High Quality Sterile IV Infusion Set Medical Equipment Surgical Equipment Disposable Light-Proof Infusion Set with CE, ISO
Factory Wholesale High Quality Sterile IV Infusion Set Medical Equipment Surgical Equipment Disposable Light-Proof Infusion Set with CE, ISO
 Butterfly Type IV Cannula Intravenous Catheter with Injection Port Infusion Needle
Butterfly Type IV Cannula Intravenous Catheter with Injection Port Infusion Needle
 Disposable Cheap Price Giving Set Infusion Set with CE Approved
Disposable Cheap Price Giving Set Infusion Set with CE Approved
 Sunton 3 Way Cannula China Arterial Cannula Supply Dressing Catheter Fixation Dressing Veca-C Sterile Medical IV Cannula/IV Catheter with Injection Port
Sunton 3 Way Cannula China Arterial Cannula Supply Dressing Catheter Fixation Dressing Veca-C Sterile Medical IV Cannula/IV Catheter with Injection Port
 Adult/Pediatric/Infant Disposable PVC Venturi Oxygen Nebulizer Breathing Aerosol Face Mask with Elastic Strap Adjustable Nose Clip Kit Set with Tubing CE/ISO
Adult/Pediatric/Infant Disposable PVC Venturi Oxygen Nebulizer Breathing Aerosol Face Mask with Elastic Strap Adjustable Nose Clip Kit Set with Tubing CE/ISO
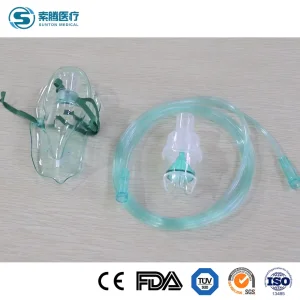 Portable Audlt/Pediatric/Infant Disposable Medical Oxygen Nasal Cannula Nebulizer Aerosal Face Facial Mask with Connecting Tubing with CE/ISO
Portable Audlt/Pediatric/Infant Disposable Medical Oxygen Nasal Cannula Nebulizer Aerosal Face Facial Mask with Connecting Tubing with CE/ISO
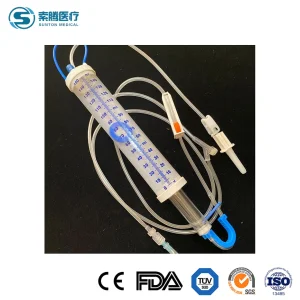 Drip Microdrip Type Sterile Intravenous 20/60 Drops Pediatric Children Adult 100/150ml Cc Disposable Burette IV Infusion Set with ISO, CE with Filter
Drip Microdrip Type Sterile Intravenous 20/60 Drops Pediatric Children Adult 100/150ml Cc Disposable Burette IV Infusion Set with ISO, CE with Filter
The global Infusion Administration Set market is witnessing unprecedented growth, driven by an aging global population and the rising prevalence of chronic diseases. As medical facilities transition toward more specialized care, the demand for precision instruments like micro-drip sets and light-proof infusion lines has surged. Currently, the market is valued at billions of dollars, with a shift towards safety-engineered devices to prevent needle-stick injuries and medication errors.
Technological convergence is the core of the 2024 industry outlook. Innovations include PVC-free and DEHP-free materials to ensure patient safety, as well as integrated precision filters that remove particulates and bacteria. We are also seeing the rise of "Smart Infusion" systems that integrate with hospital networks for real-time monitoring, ensuring that high-potency drugs are delivered with 100% accuracy in clinical environments.
International medical distributors and hospital GPOs (Group Purchasing Organizations) are no longer just looking for the lowest price. Their core requirements now emphasize regulatory compliance (CE, ISO 13485), supply chain resilience, and OEM flexibility. There is a growing need for "localization-ready" products that can adapt to different medical standards in Europe, North America, and Southeast Asia.
The following manufacturers represent the pinnacle of medical engineering, quality control, and global distribution. These companies define the standards for intravenous therapy worldwide.
A global leader headquartered in the USA, setting the gold standard for infusion safety and vascular access technologies.
A German powerhouse known for high-end infusion therapy, clinical nutrition, and comprehensive hospital supply chains.
Specializing in critical care and hospital products, Baxter is a key innovator in smart infusion pump integration.
Following the acquisition of Smiths Medical, they are a primary provider of IV therapy products and infusion systems.
Japan’s leading medical device manufacturer, focusing on high-precision needles and infusion technology for delicate procedures.
A specialized leader since 1996, bridging the gap between high-capacity manufacturing and premium quality. ZNG is renowned for its vast range of specialized needles and customized infusion sets.
Highly regarded for its dialysis and infusion product lines, balancing Japanese engineering with global manufacturing scale.
A leader in infusion, transfusion, and clinical nutrition, particularly dominant in the European and Asian markets.
Focused on specialized enteral and parenteral infusion pumps and sets, catering to home healthcare sectors.
Pioneers in syringe pumps and specialized infusion sets for oncology and pain management.
Zhejiang ZNG Medical founded in 1996, as the leading medical device company, we are dedicated to the researching and developing and producing of injection products. Through the ISO 9001 quality system and CE product certification, we provide secure and efficient products to the global market.
Our specialization in needle technology is second to none. We offer:
Mission: We are committed to public health by listening to customer requests, allowing us to continuously improve existing products and innovate new medical solutions.
Requirement Analysis & Quotation with Free Samples
PO/PI Confirmation and Deposit Processing
Advanced Production with Photo Updates & QC
Global Logistics, Original Docs & After-sales
 Sterilized by EO
Sterilized by EO
In critical care, the need for titration infusion sets with burettes is paramount. Our sets allow for micro-precise delivery of life-saving medications where every drop counts.
Children require smaller volumes and slower delivery rates. Our 60-drop micro-drip sets and 100ml/150ml burettes ensure maximum safety for the youngest patients.
Many oncology drugs are light-sensitive. We provide Light-Proof Infusion Sets that protect the integrity of the medication throughout the delivery process.
Portable, robust, and sterile. Our IV giving sets are designed to function reliably in high-pressure environments, from ambulances to remote field hospitals.
Choosing a top-tier Chinese factory like ZNG Medical offers a unique blend of efficiency and innovation.
From stainless steel cannula production to final EO sterilization, we control the entire process. This reduces lead times and ensures consistency that trading companies cannot match.
We don't just sell off-the-shelf products. Our engineering team specializes in ODM/OEM projects, creating custom needle gauges or set configurations tailored to specific surgical needs.
With CE and ISO certifications, our manufacturing standards align with international medical device regulations, ensuring easy market entry for our partners in Europe and America.
 Sunton China 110 Electrode Piece Manufacturers Free Sample Medical Disposable ECG Electrode Slice
Sunton China 110 Electrode Piece Manufacturers Free Sample Medical Disposable ECG Electrode Slice
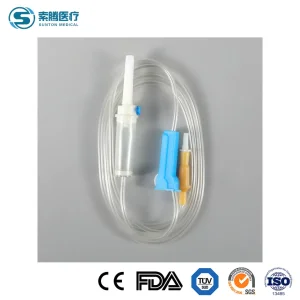 Disposable Sterile IV Medical Ordinary Luer Slip/Lock Infusion Set with Needle CE, ISO with Filter Intravenous Drip Chamber Type
Disposable Sterile IV Medical Ordinary Luer Slip/Lock Infusion Set with Needle CE, ISO with Filter Intravenous Drip Chamber Type
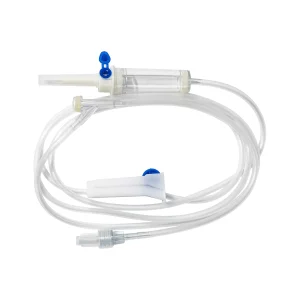 IV Giving Sets IV Set Disposable Infusion Set
IV Giving Sets IV Set Disposable Infusion Set
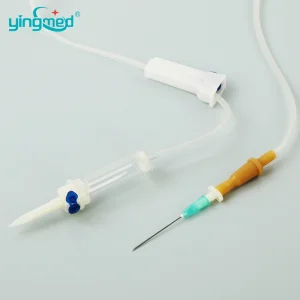 Disposable Medical Infusion Set IV Set
Disposable Medical Infusion Set IV Set
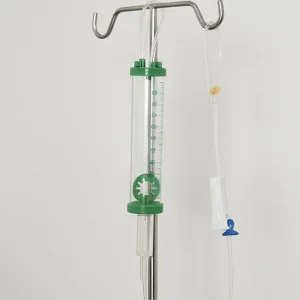 High-Quality OEM/ODM 100ml Burette Infusion Set for Medical Use Quality Medical Infusion Set Designed for Intravenous Applications
High-Quality OEM/ODM 100ml Burette Infusion Set for Medical Use Quality Medical Infusion Set Designed for Intravenous Applications
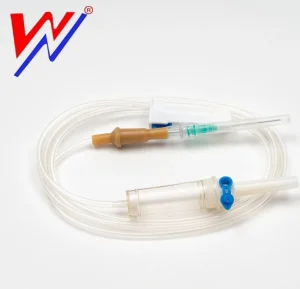 IV Infusion Set with High Quality and Competitive Price and Double Chamber
IV Infusion Set with High Quality and Competitive Price and Double Chamber
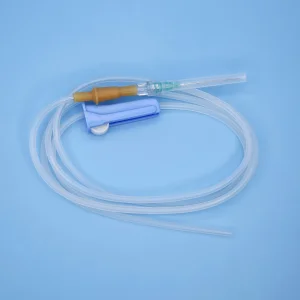 Infusion Set IV Set IV Giving Set with Needle with CE/ISO/FDA
Infusion Set IV Set IV Giving Set with Needle with CE/ISO/FDA
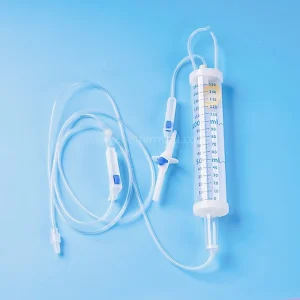 Bm® Disposable High Quality Medical Sterile Burette Infusion Set ISO/CE
Bm® Disposable High Quality Medical Sterile Burette Infusion Set ISO/CE