Basic Information
Transport Package
Box+Carton
Trademark
OBM/OEM
Origin
China
HS Code
3002150090
Production Capacity
5,000,000 Pieces/Month
Product Description
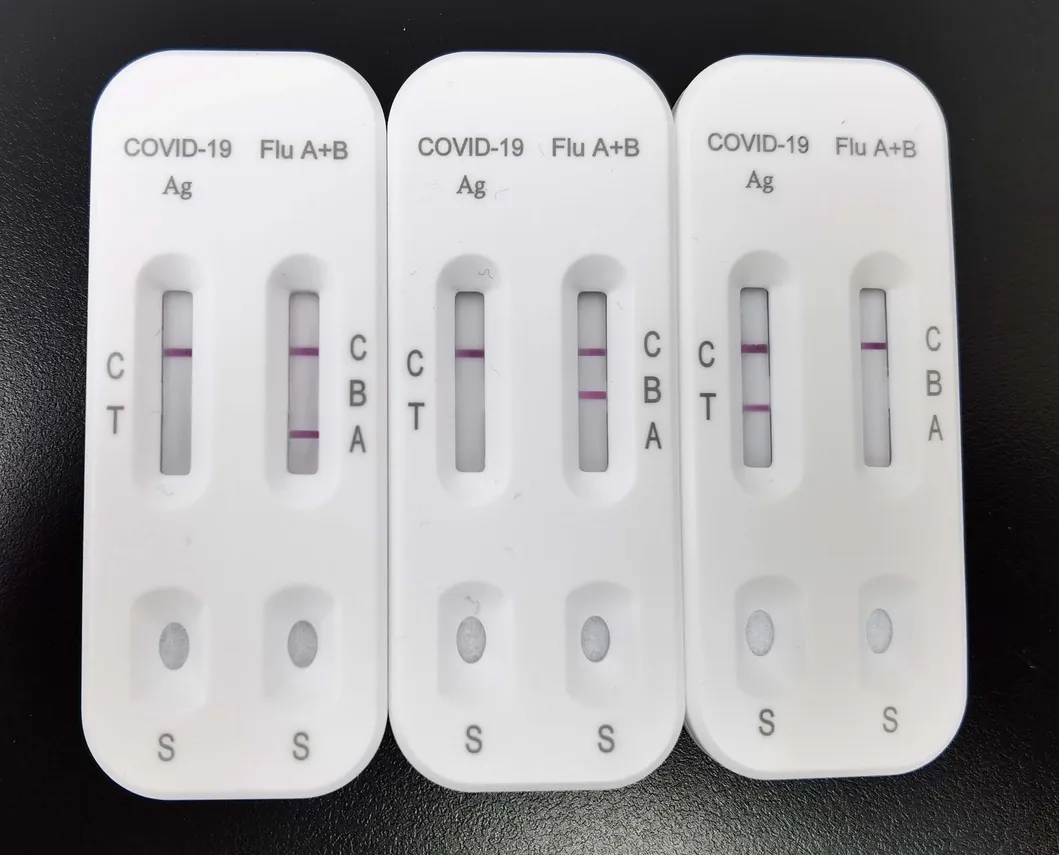
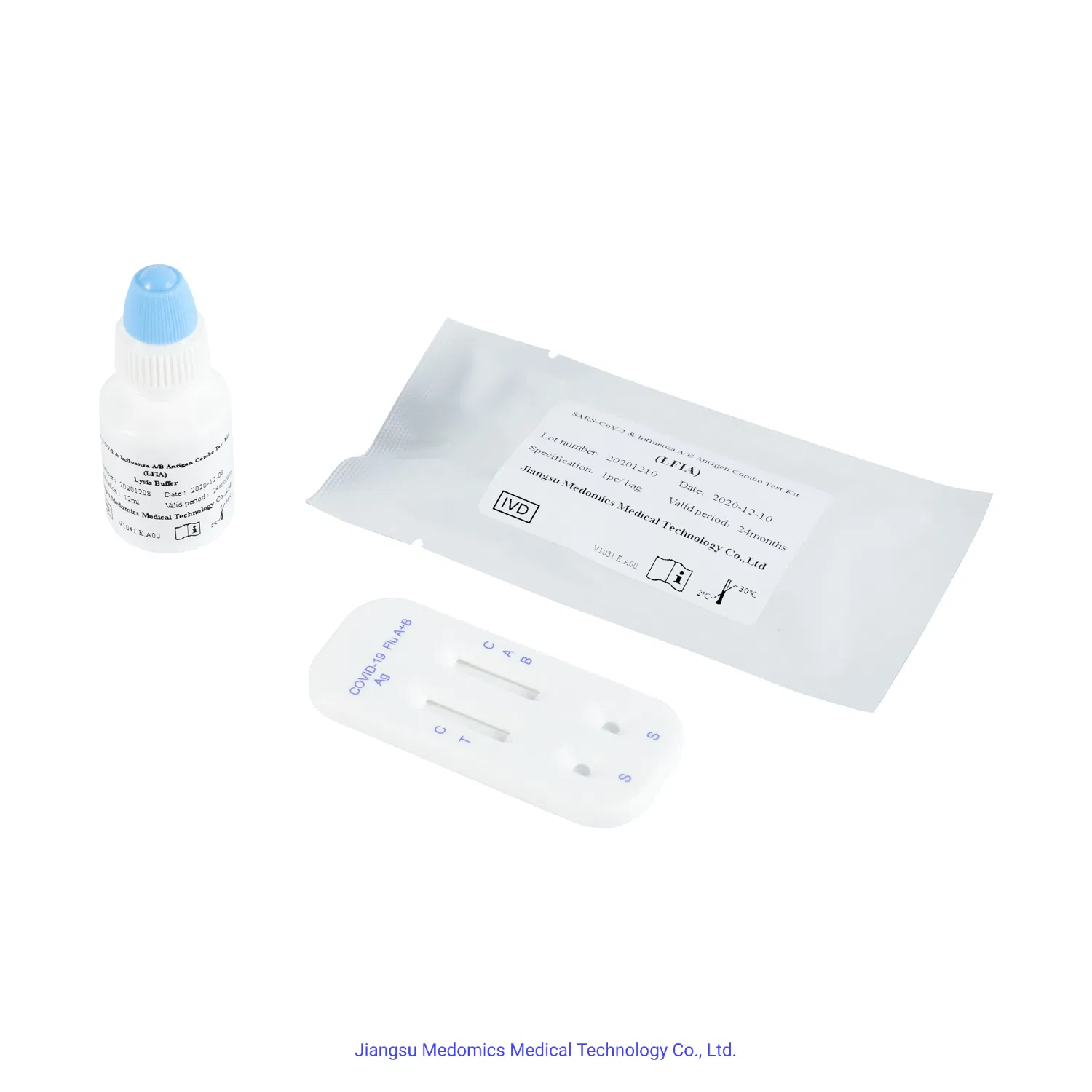
Flu A&B and Novel Virus Antigen Test Kit (LFIA) is designed for the qualitative detection of influenza virus type A, influenza virus type B, and novel virus antigens in human samples in vitro. Since influenza and the novel virus present similar symptoms and infection routes, this combined test kit serves a critical clinical role in rapid differential diagnosis.
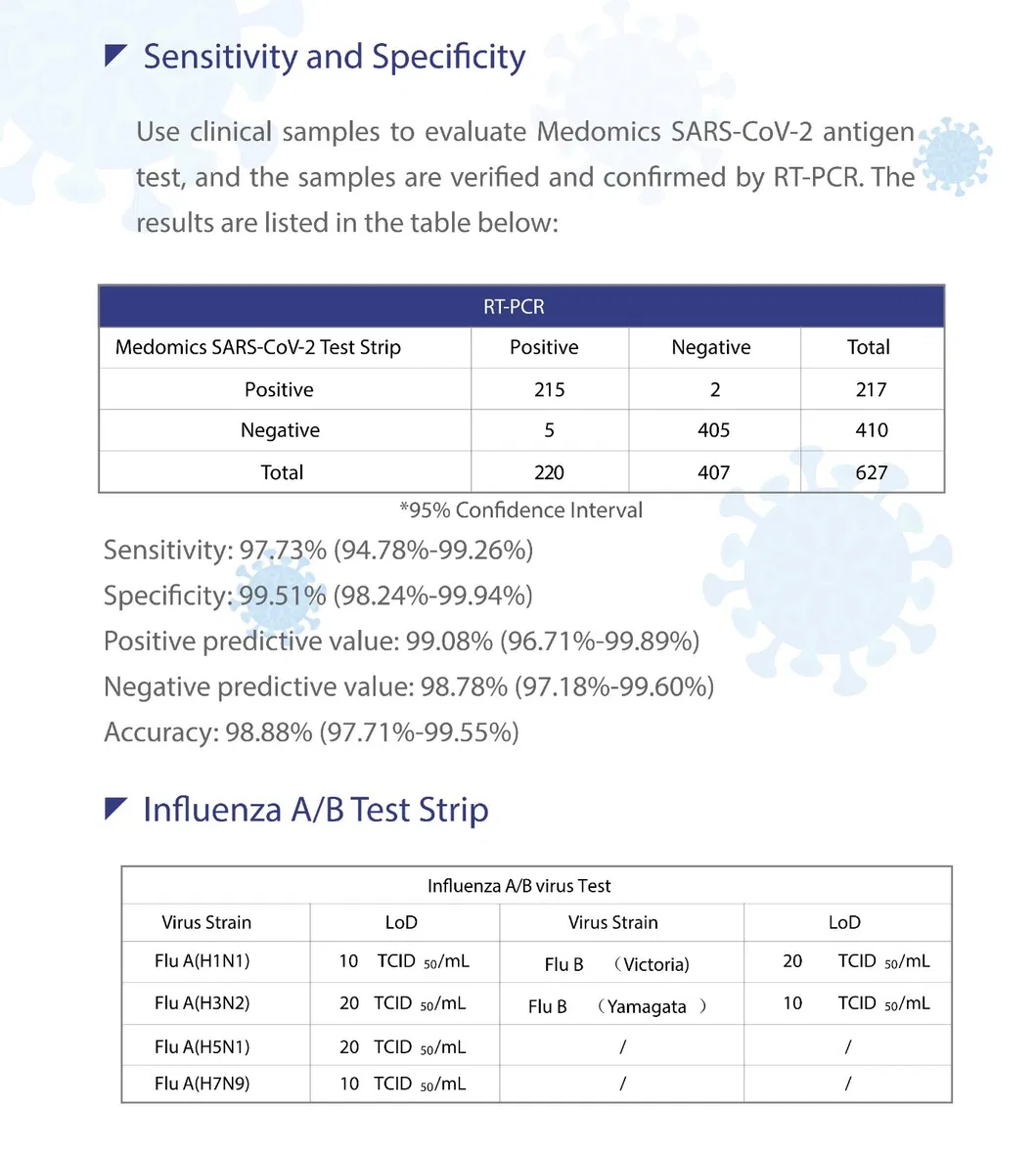
- Sensitivity: >95%
- Specificity: 100%
- Reaction Time: 10 minutes
- Sample Type: Nasopharyngeal swab
- Storage Temperature: 4-30ºC
Usage Instructions & Details


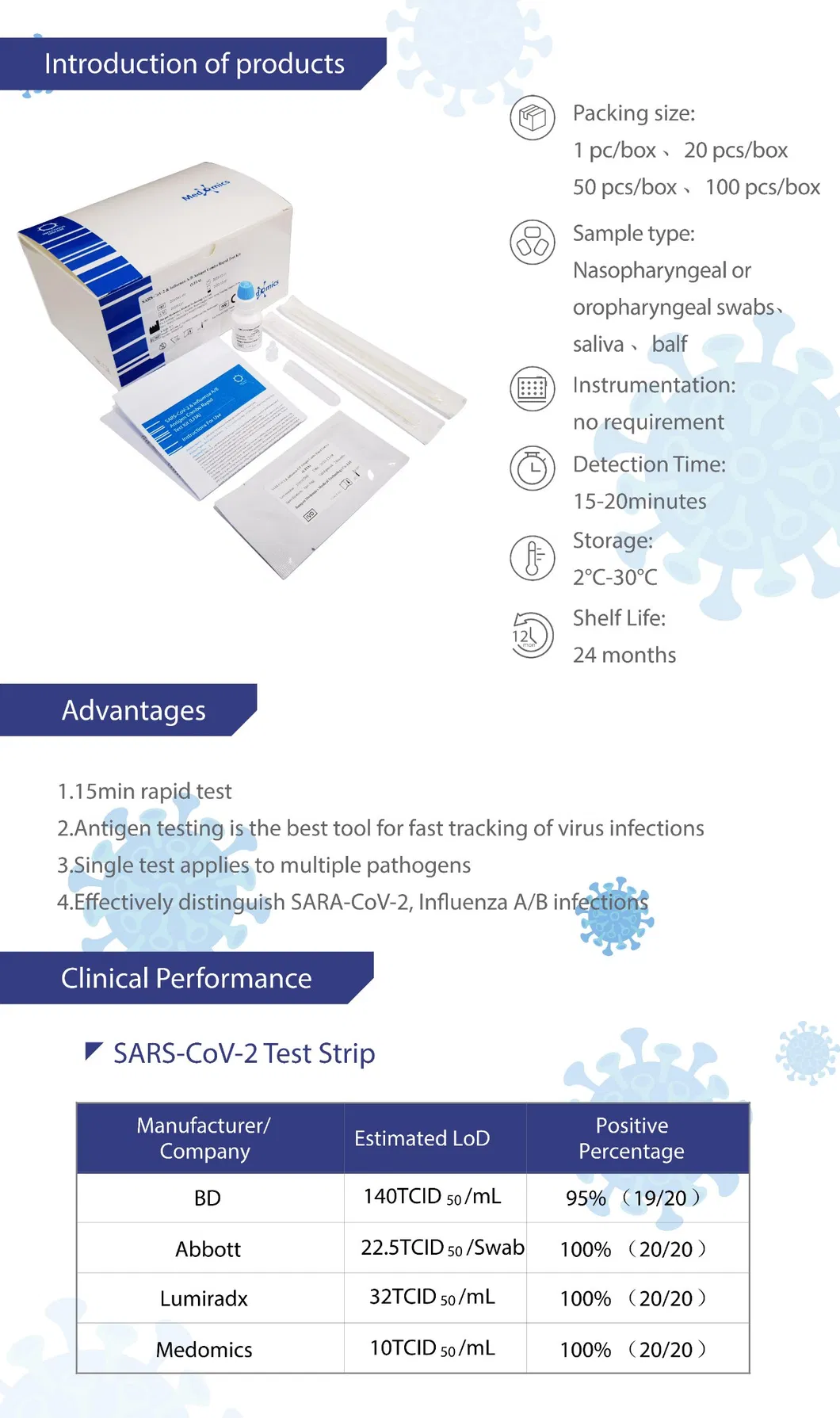
About the Manufacturing Facility

The manufacturer is an international high-tech enterprise driven by innovation in medical device R&D, production, and sales. Focusing on the diagnosis of microorganisms, tumors, and rare diseases, the facility manages a total of more than 10,000 square meters of GMP purification production workshops that meet global standards (FDA, CE, and CFDA). With multiple R&D centers globally, the technical team includes experts in immunology, microbiology, and applied physics from renowned international institutions.


 ZNG Medical
ZNG Medical