


 ZNG Medical
ZNG Medical











(Colloidal Gold Method)

| Type | Product Name | Catalog No. | Specimen | Format | Test/Kit | Cut-off (ng/mL) |
|---|---|---|---|---|---|---|
| DOA Single-Drug | AMP Amphetamine | DAM-101/102 | Urine | Strip/Device | 50T/40T | 1000 |
| BAR Barbiturates | DBA-101/102 | Urine | Strip/Device | 50T/40T | 300 | |
| BUP Buprenorphine | DBU-101/102 | Urine | Strip/Device | 50T/40T | 10 | |
| BZO Benzodiazepines | DBZ-101/102 | Urine | Strip/Device | 50T/40T | 300 | |
| COC Cocaine | DCO-101/102 | Urine | Strip/Device | 50T/40T | 300 | |
| FTY Fentanyl | DFT-101/102 | Urine | Strip/Device | 50T/40T | 100 | |
| KET Ketamine | DKE-101/102 | Urine | Strip/Device | 50T/40T | 1000 | |
| MET Methamphetamine | DME-101/102 | Urine | Strip/Device | 50T/40T | 1000 | |
| MOP Morphine 300 | DMO-101/102 | Urine | Strip/Device | 50T/40T | 300 | |
| THC M-arijuana | DTH-101/102 | Urine | Strip/Device | 50T/40T | 50 | |
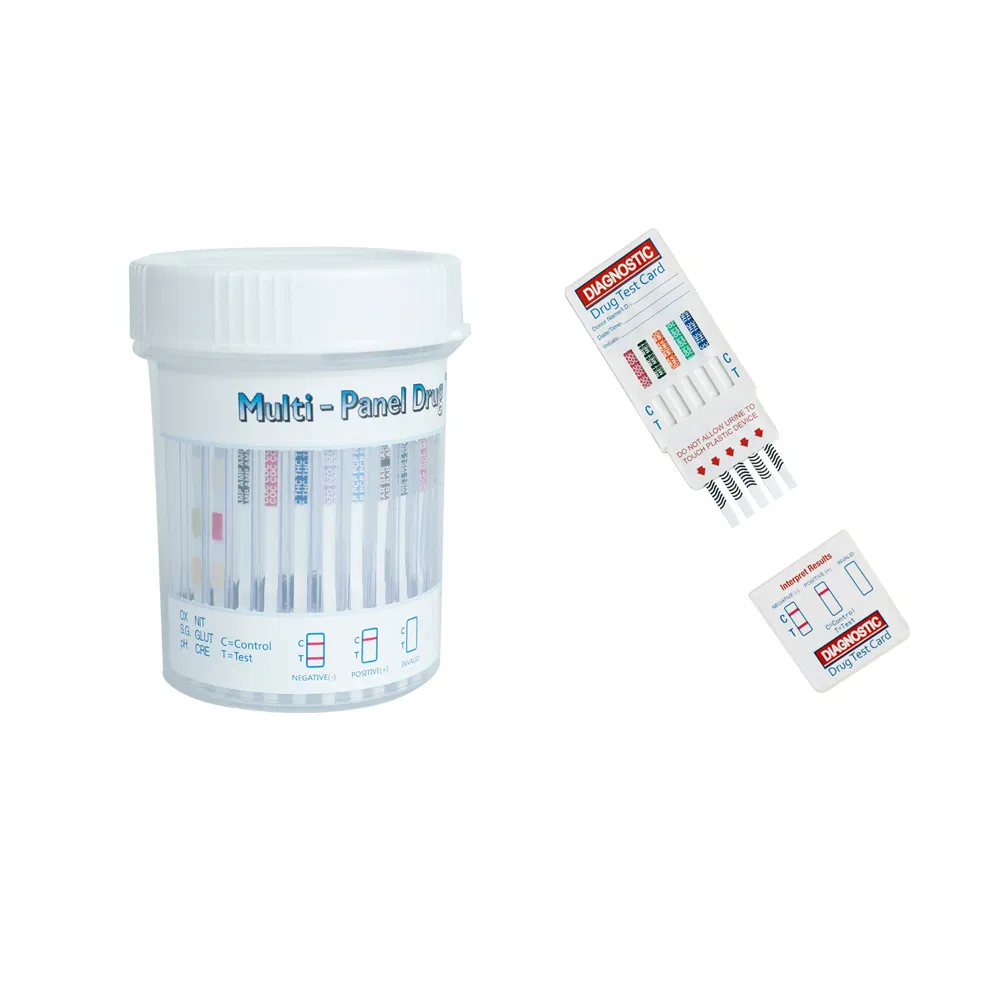
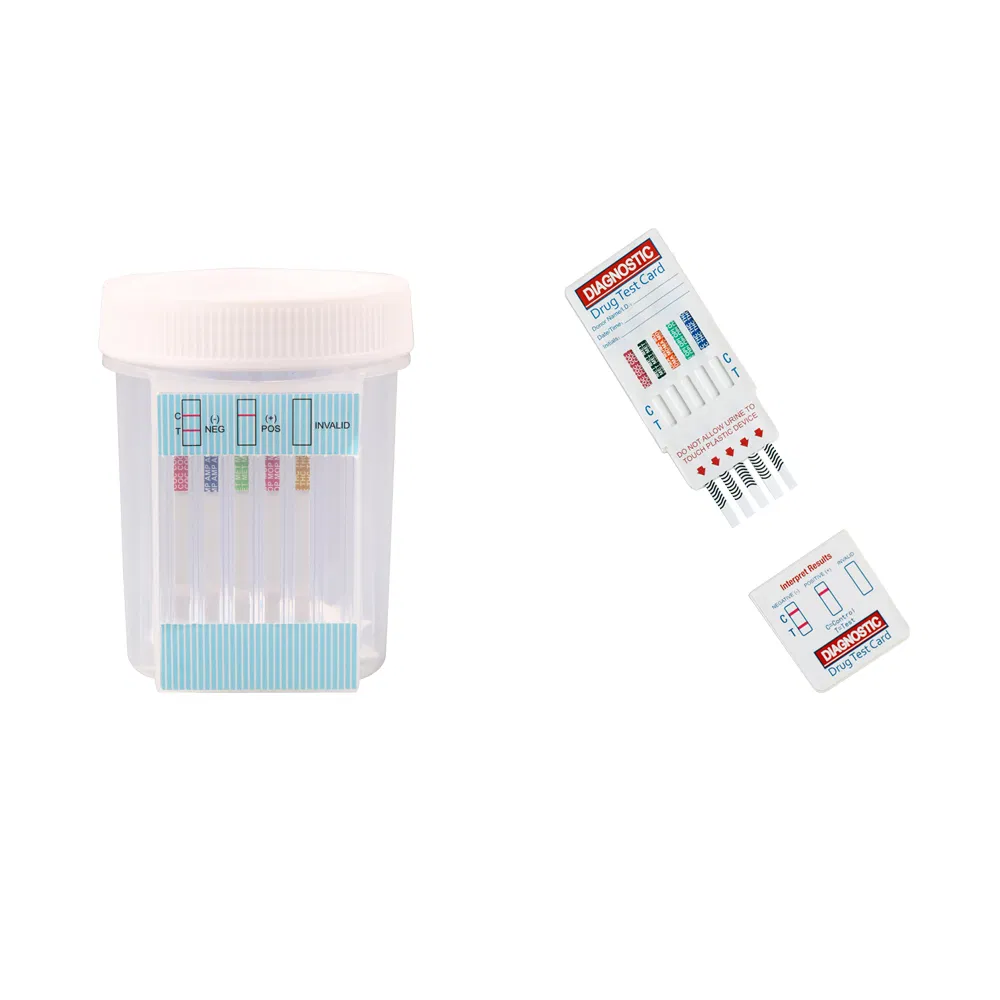
| Multi-Drug Panel | Multi-Drug One Step Panel | DOA-124 to 1124 | Urine | 2-12 Drugs | 20 Test/box | - |
| Integrated E-Z Split Cup | DBO-127 to 1127 | Urine | 2-12 Drugs | 20 Test/box | - |
The Multi-drug One Step Test Kit (Colloidal Gold Method) is a rapid chromatographic immunoassay for the qualitative detection for the drugs and drug metabolites in human urine. The detection limits cut-off concentrations are specified in the product documentation.
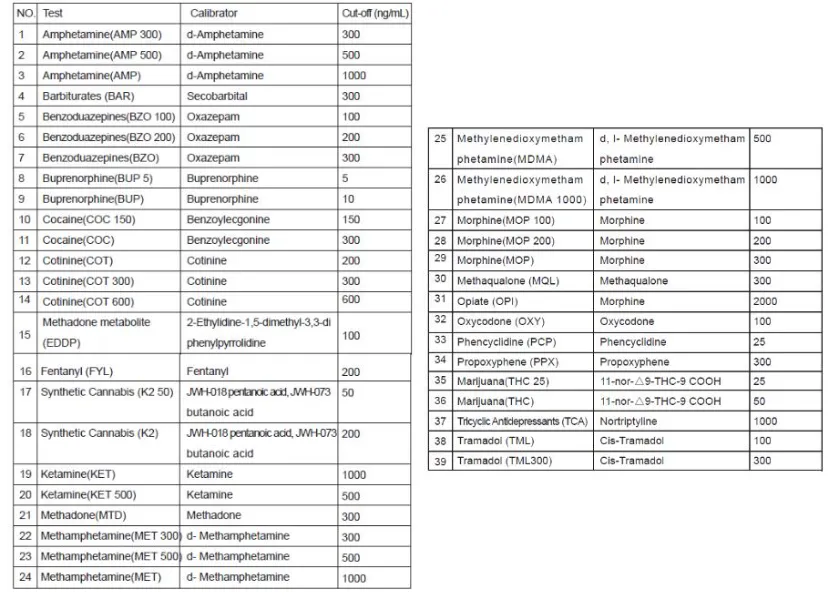

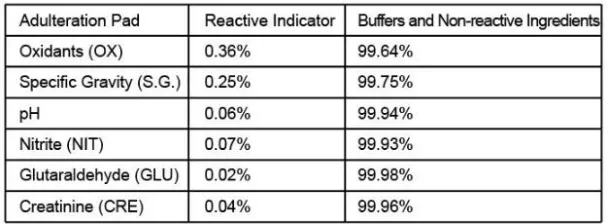
Configurations of multi-drug test kit come with any combination of drug analytes with or without specimen validity test (S.V.T.). This assay provides only a preliminary analytical test result. A more specific alternative chemical method must be used to obtain a confirmed analytical result. Gas chromatography/Mass spectrometry (GC/MS) is the preferred confirmatory method.
Equilibrate the tests, urine specimens, and/or controls to room temperature (15-25ºC) prior to testing.
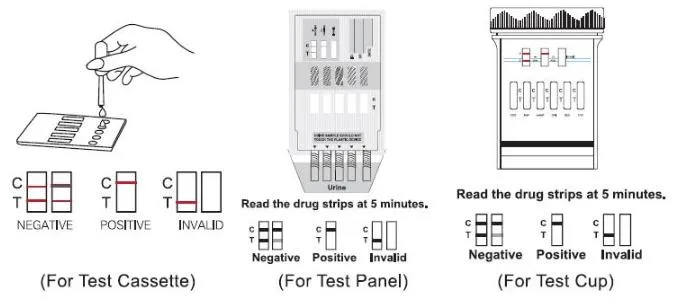
Negative: Two distinct colored lines appear. One line in control (C) and one in test (T).
Positive: Only one colored band appears in control (C). No line in test (T).
Invalid: No line appears in the control region (C).



