1 / 4
 ZNG Medical
ZNG Medical








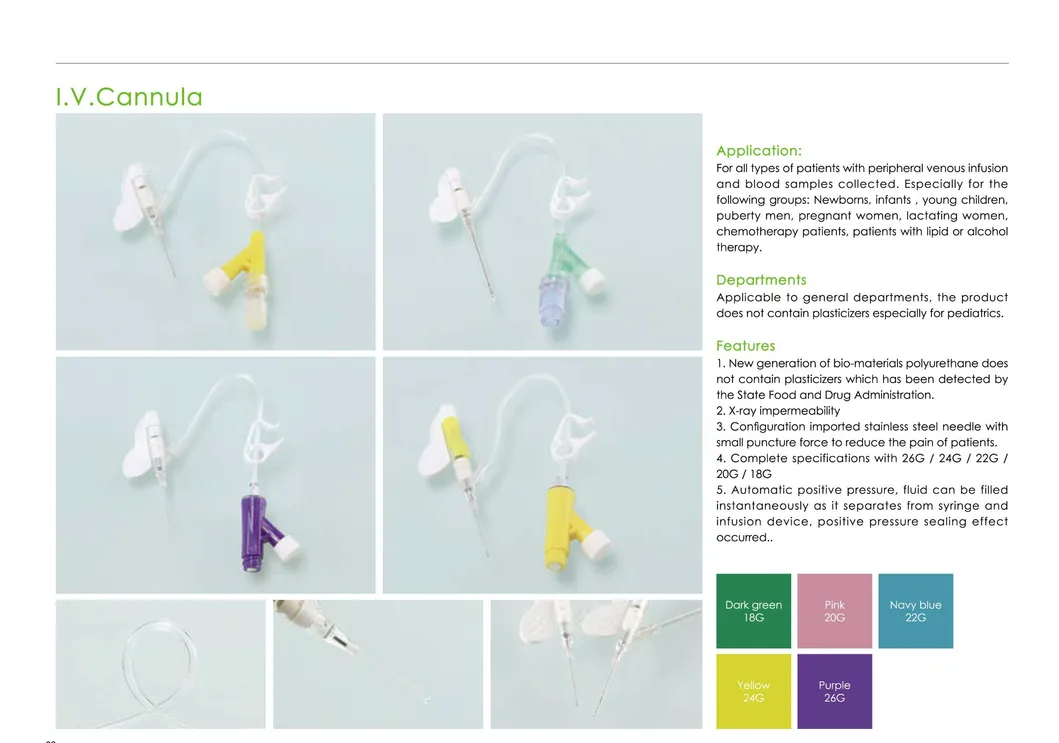
Application:
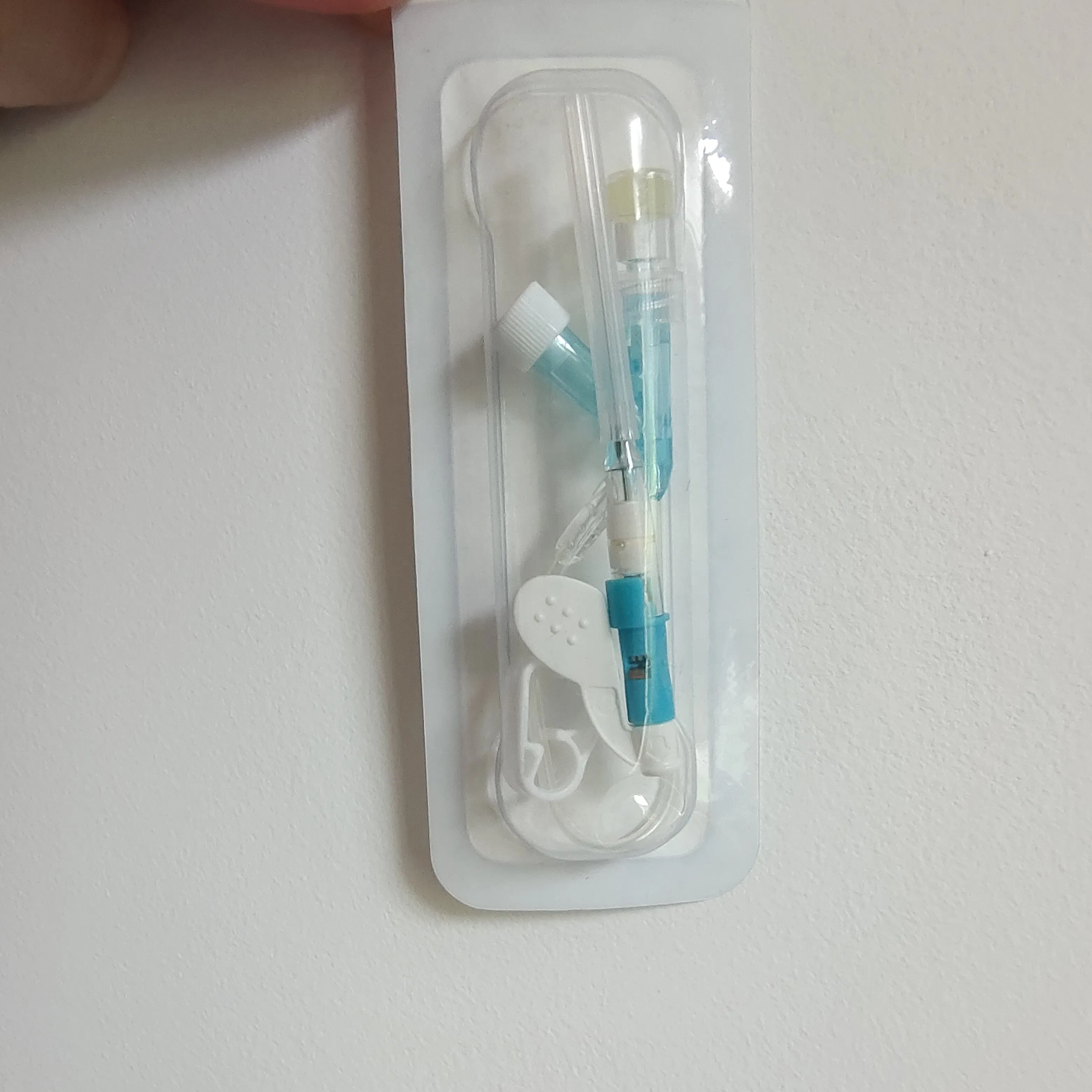
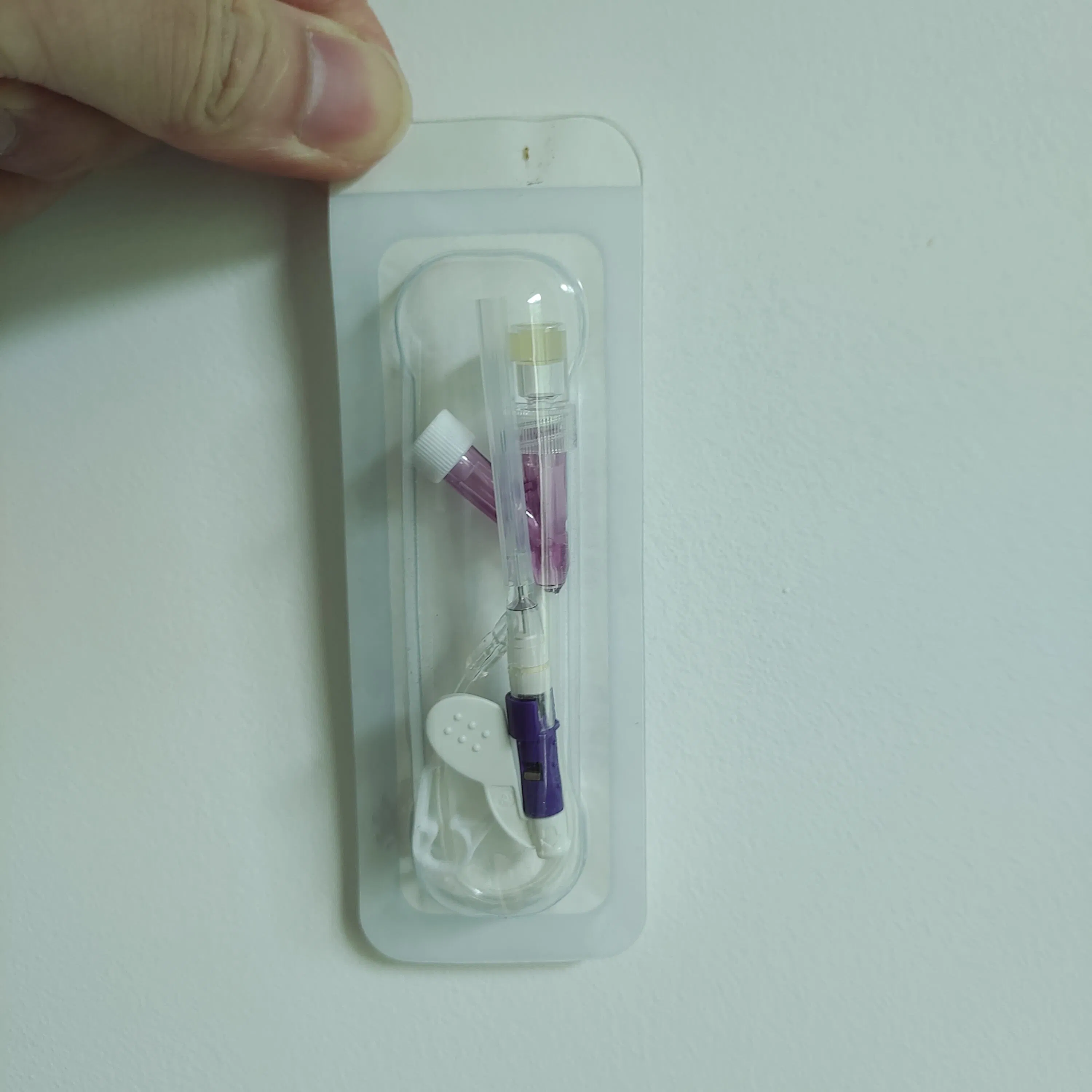
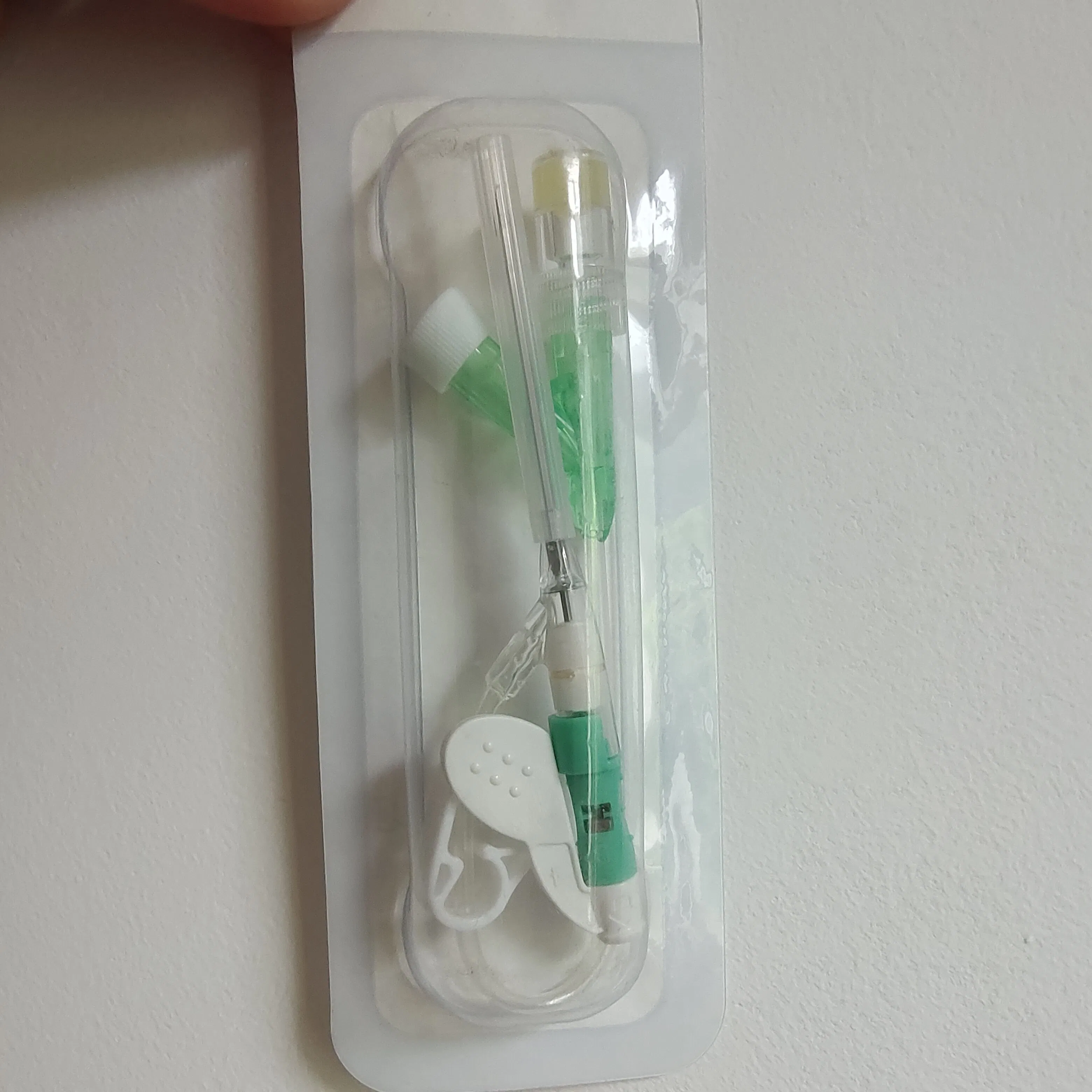
Designed for all types of patients requiring peripheral venous infusion and blood sample collection. It is especially suitable for sensitive groups including newborns, infants, young children, pregnant women, chemotherapy patients, and patients undergoing lipid or alcohol therapy.
Clinical Departments:
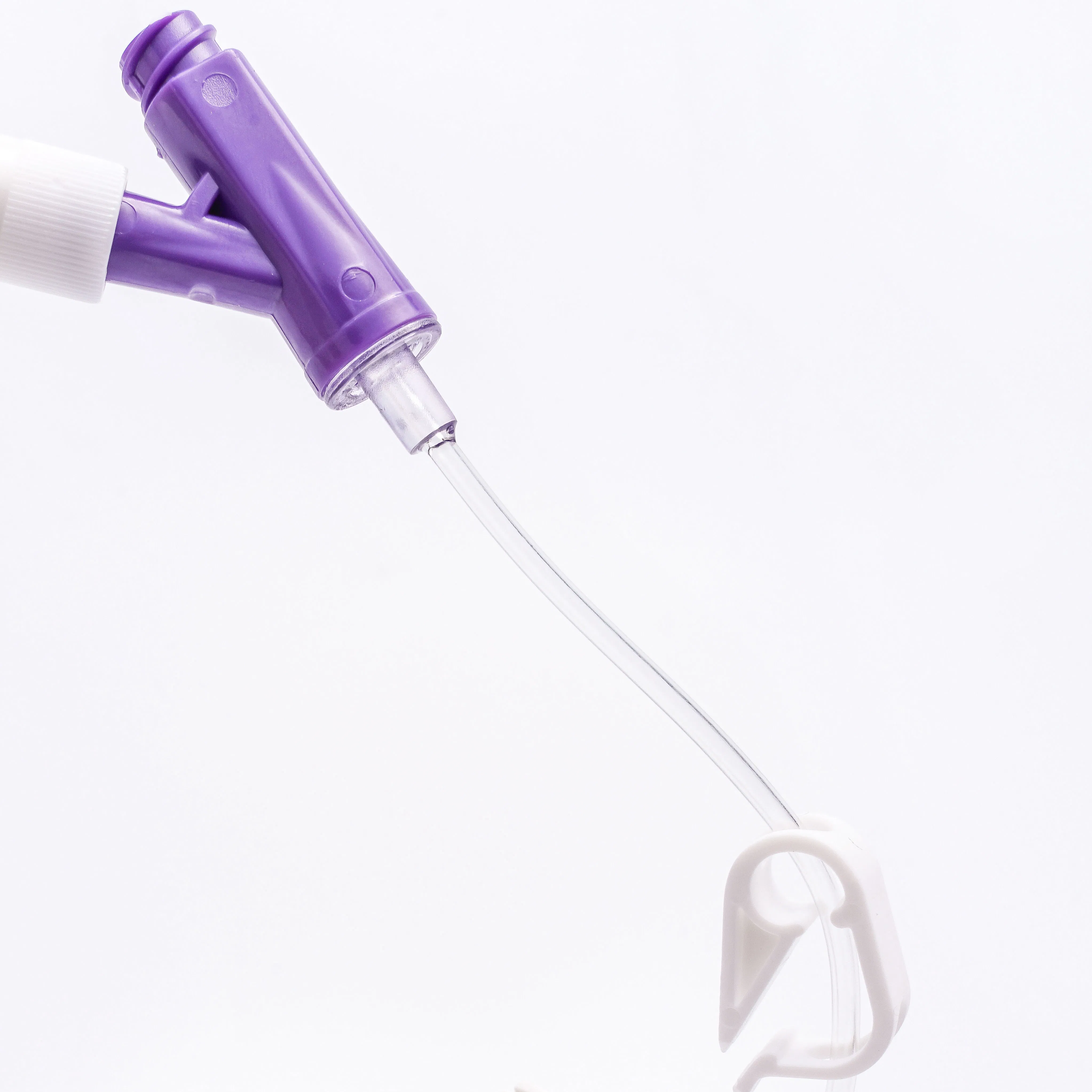
Applicable to general medical departments. The product is DEHP-free, making it particularly safe for pediatrics.
Key Features:
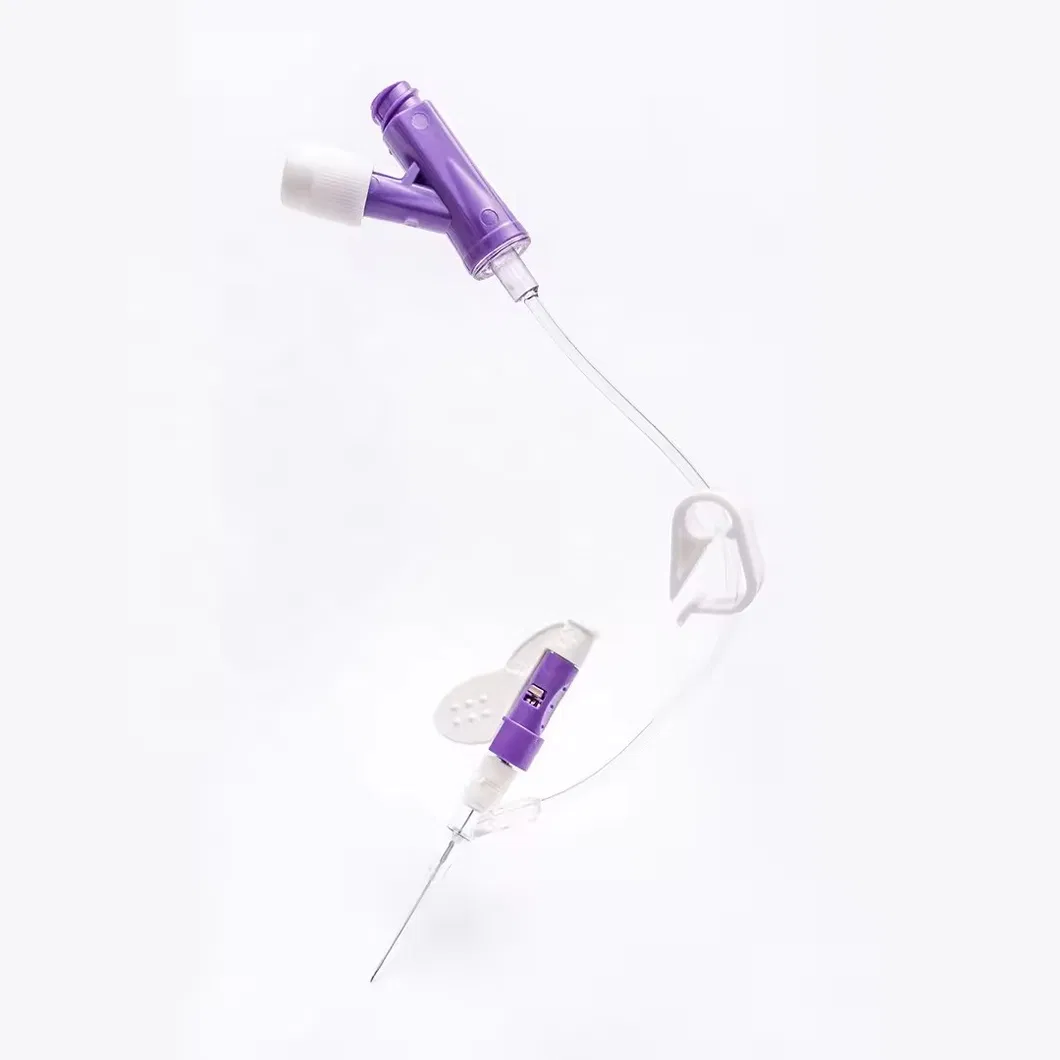
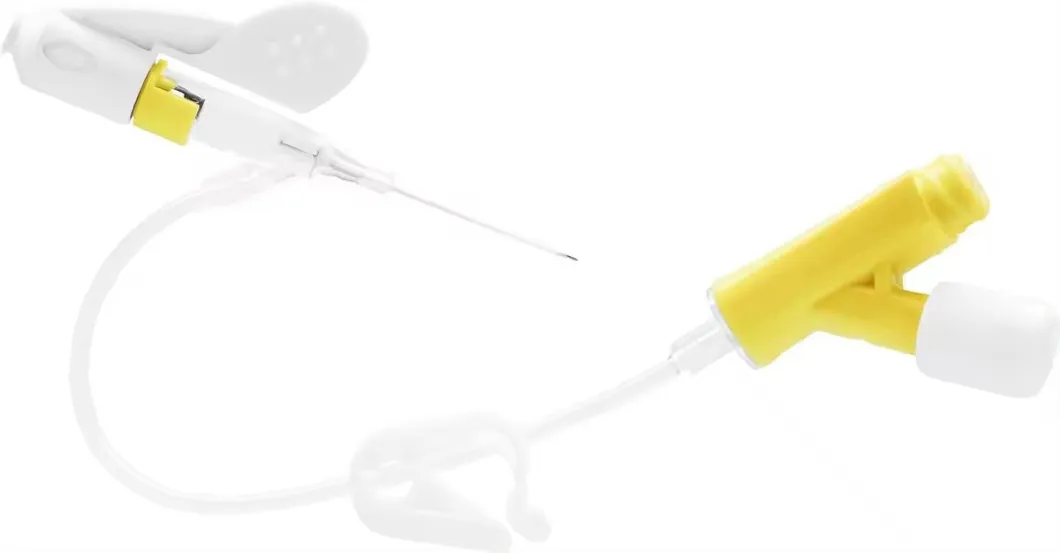
| Product Code | Specification | Length | Type | Color | Patient Type |
|---|---|---|---|---|---|
| 6321842 | 18G | 30mm | Double Safety | Green | Adult |
| 6251842 | Normal | Green | |||
| 6322042 | 20G | 30mm | Double Safety | Pink | |
| 6262034 | 25mm | Positive Pressure | Pink | ||
| 6322231 | 22G | 25mm | Double Safety | Blue | |
| 6252232 | Normal | Blue | |||
| 6322422 | 24G | 19mm | Double Safety | Yellow | Adult/Children |
| 6322612 | 26G | 14mm | Double Safety | Purple | Children |
Focus on quality, Respect on life!